Getting sick is never fun, and it always seems to happen at the most inconvenient times. But fear not! When… Read Full Article
Making health care decisions can be daunting which is why we employ a comprehensive, team-based approach so our patients feel… Read Full Article
Have you heard about friends and neighbors getting sick from the “stomach flu?” This is usually not from influenza, but… Read Full Article
Get ready for the new year with a renewed focus on your health. Our Hoag Medical Group physicians are here… Read Full Article
As a pediatrician, I see first-hand the rollercoaster of emotions parents go through regarding the recent emergency use approval of… Read Full Article
It is that time of the year… time to get your flu vaccine! It may be more important than ever to get… Read Full Article
This is an exhilarating time for children and parents as we head back to school to resume in-person learning. Our… Read Full Article
This is an exhilarating time for children and parents as we head back to school to resume in-person learning. Our… Read Full Article
After more than a year spent quarantining in our homes, people are starting to open their minds (if not their… Read Full Article
She was in tears. She was 16 years old. She had just told me she did not want to live… Read Full Article
For Father’s Day, some dads get cards or homemade gifts. This Father’s Day, consider getting a health screening including a… Read Full Article
This is a difficult time for everyone – parents and children alike. With mental health concerns on the rise, it… Read Full Article
An integral part of my practice is working with my patients to optimize their health, helping them to eat better,… Read Full Article
During this challenging and unprecedented time, there is one thing to know – Hoag Medical Group (HMG) is here for… Read Full Article
For the first time in almost 30 years, the influenza B is dominating flu season. This is particularly worrisome for… Read Full Article
The sight of a newborn baby fills most of us with a strong desire to protect. We swaddle against the… Read Full Article
Choosing your primary care physician has the potential to be one of the most important decisions you will make. Typically,… Read Full Article
Are you regretting that decision to help a friend throw away that Plasma TV? Chances are you and 84% of… Read Full Article
“So, how was school today?” “Fine.” “What did you do?” With shoulders shrugged, “I don’t know.” This is one… Read Full Article
Last month I got a frantic phone call from a parent who had just found out that her daughter, who… Read Full Article
When it comes to traveling with young children, parents often sweat the small details – shoving spare diapers into every… Read Full Article
At nearly every visit to your physician, one of the initial things you’ll experience is a blood pressure screening. This… Read Full Article
Summer is here, an enjoyable time for families, full of adventure, sun and outside activities. Unfortunately, as exciting as it… Read Full Article
For thousands of years, we have understood that “food is medicine;” what goes into our bodies has a significant effect… Read Full Article
Osteoporosis affects more than 50 million Americans and found in all races and gender. Osteoporosis is not part of the… Read Full Article
What are you doing at exactly this moment? What is going on with your body? What is happening with your… Read Full Article
We have all been hearing about the recent rise of vaccine preventable illnesses in the news. Of particular concern is… Read Full Article
When a patient is using five or more medications it is defined as “polypharmacy.” Polypharmacy is more common in older… Read Full Article
Chicken pox parties were popular in the pre-vaccine era as a way to make sure your child was infected with… Read Full Article
Your head is throbbing, the world is thrumming, maybe you feel nauseous or faint. When headaches strike, it can be… Read Full Article
Parenting is an exceptional and exciting experience. Thankfully, it is not a solo one. In addition to the cast of… Read Full Article
We can never predict when an injury is likely to occur, but with the right preparation and precautions, we may… Read Full Article
Shopping malls are crawling with snifflers, airports are bustling with coughers, and everywhere we turn we’re confronted with viruses that… Read Full Article
When you are trying to stay on a healthy course with a balanced diet, the holidays can feel daunting. Candy… Read Full Article
For many of us, “the most wonderful time of the year” is anything but. If we let the holiday season… Read Full Article
When that little light goes off in your car, you know it’s time for an oil change. Unfortunately, your body… Read Full Article
People generally understand what type 2 diabetes is. But a diagnosis of pre-diabetes tends to elicit one of two reactions:… Read Full Article
The number of children diagnosed with autism spectrum disorder (ASD) is on the rise. The Centers for Disease Control and… Read Full Article
Most people don’t consider Thanksgiving to be the healthiest of holidays. Between the ladles of gravy and the piles of… Read Full Article
Many parents come to me with questions about what car seat is best for their child. This is an extremely… Read Full Article
The only thing more contagious than the flu is misinformation. Every year, when I remind my patients to get their… Read Full Article
When bringing home your newborn bundle of joy from the hospital, you might be wondering why their skin appears yellow.… Read Full Article
After a long day, many of us want to come home and relax in front of the TV. But that… Read Full Article
It may be difficult to accept the diagnosis of hypertension or high blood pressure and even harder for some people… Read Full Article
Whether your child has spent the summer as a zombie in front of a glowing screen or as a blue-lipped… Read Full Article
More people are diagnosed with skin cancer each year, than all other types of cancer combined. If that statistic startles… Read Full Article
The first rule of potty training is to not fear potty training. It will likely be messy. It probably won’t… Read Full Article
You have probably heard that to eat healthier, you should cook your meals at home. That’s a fantastic piece of… Read Full Article
We all admire kids who seem to do it all. The Ivy League-bound star athlete and academic decathlon champ whose… Read Full Article
Often times my patients come into the office to discuss their challenges with sleep. Here are some of the things… Read Full Article
When we get ready to travel, we plan our itinerary and pack all of the necessities, but do we create… Read Full Article
You need to chill out. Seriously. Doctor’s orders. Research has long established a link between anxiety and health issues as… Read Full Article
Most people who have Hepatitis C don’t even know they have it. Hepatitis C can damage the liver and lead… Read Full Article
Generally speaking, fevers are not dangerous. While they are uncomfortable, most fevers that parents encounter are manageable and benign. Even… Read Full Article
Babies go through an astounding amount of changes during their first year of life. From learning to crawl to hearing… Read Full Article
We think of high cholesterol as the punishing conclusion of many years of bad habits. Unfortunately, however, even young children… Read Full Article
Fatigue is a common issue for many people, and as a physician, I often hear this in my practice. Finding… Read Full Article
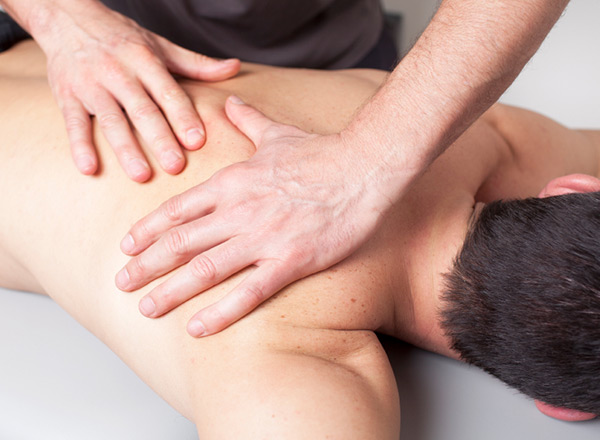
In the past, when I’d tell people that I practice sports medicine, the first thing they’d ask me is, “What’s… Read Full Article
If you’re waiting until you’re sick to see your doctor, you’re waiting too long. Preventive medicine is important, and annual… Read Full Article
I have a secret diet that works for all children and families to ensure a healthy balance of fruits, vegetables,… Read Full Article
You’ve been enjoying your baby’s “tooth-less” smiles for a few months and now there are some tiny little white teeth… Read Full Article
The field of Rheumatology is a subspecialty of Internal Medicine and Pediatrics that focuses on the diagnosis and therapy of… Read Full Article
Q: What is the best kind of diet to lose weight? A: Patients ask me all the time what diet… Read Full Article
Do we experience more mental health disorders during the holidays? Not really. The popular belief that rates of clinical mental… Read Full Article
As a Rheumatologist, I often have to explain to patients what it means to have an autoimmune disease. To understand… Read Full Article
Medications allow us to overcome illness, power through pain and transform some fatal diseases into merely chronic ones. But no… Read Full Article
Vitamin D is constantly making headlines. Are we getting enough? Are we getting too much? Do we need to take… Read Full Article
People are familiar with two camps of thoughts regarding childhood vaccinations: Those who avail themselves of the scientific advances that… Read Full Article
Fibromyalgia can be a daunting diagnosis for some individuals. Most people think it’s an inflammatory condition or an autoimmune condition… Read Full Article
If you eat fewer calories than you burn off, you will lose weight. It’s that simple. But for many people,… Read Full Article
There is a lot of news these days about gluten. Whether it is a gluten intolerance, gluten allergy, or gluten… Read Full Article
There’s nothing that pulls at your heartstrings more than seeing your child sick or injured. At some point, childhood sickness… Read Full Article
There are over 100 different types of arthritic conditions and the management and treatment is vastly different from one to… Read Full Article
While the term Attention Deficit Disorder (ADD) is commonly associated with children, it is also a condition that affects many… Read Full Article
Living in Southern California, we are exposed to sunshine nearly year-round. The warm weather makes it ideal for enjoying outdoor… Read Full Article
Grownups might not think twice about taking over-the-counter (OTC) drugs to ease the symptoms of a cold, but cough and… Read Full Article
Research shows that people who continue to maintain close friendships and find other means to interact socially live longer than… Read Full Article
Every so often, medical research produces an unexpected finding that causes a collective response of, “Whoa!” Case in point: In… Read Full Article
The two pillars of good health are taking care of oneself with healthy lifestyle choices and undergoing appropriate screening exams.… Read Full Article
That stinging, burning, often annoying sensation you may get when trying to urinate – what is it? There can be… Read Full Article
Exhortations to keep our kids physically active on a daily basis — that is, to get exercise — have become… Read Full Article
You should not eat sugary foods. It’s that simple. Our bodies don’t need them, our teeth don’t want them, and… Read Full Article
For the majority of people, vitamin supplementation is not necessary. Adequate vitamin intake can usually be attained by a healthy… Read Full Article
For young families, water represents a challenging paradox: It can provide hours of enjoyable, carefree fun, but it can also… Read Full Article
When we get ready to travel, we plan our itinerary and pack all of the necessities, but do we create… Read Full Article
A Medical Geneticist is a physician that undergoes additional training and education to become board certified in genetics. This physician… Read Full Article
As a parent, the only thing worse than catching the flu yourself is watching as one of your children suffers… Read Full Article
The song, “Don’t Worry Be Happy,” often makes people smile. Does this simple phrase provide benefit for healthy aging? Negative… Read Full Article
Maybe you’re familiar with the saying, “Life is not measured by the number of breaths we take, but by the… Read Full Article
Ever wish you could go grocery shopping with your doctor and seek their advice? St. Joseph Hoag Health recently launched… Read Full Article
As a parent of a young child, it may feel like you’re on a never-ending cycle of wiping runny noses,… Read Full Article
The holidays can be brutal on anyone’s efforts to maintain and attain ideal weight. Something purposeful YOU can resolve to… Read Full Article
Head trauma in childhood is terrifying – and sadly, common. Each year, 62,000 children experience brain injuries requiring hospitalization. Assessing… Read Full Article
When a temper tantrum strikes, especially in public, a parent’s natural response may be to console and do whatever they… Read Full Article
The holiday season is when childhood memories are made: Parties! Presents! Sugary treats! But the season also brings a break… Read Full Article
We all know that airplanes are filthy. Train stations are thick with coughs and sneezes, and bus terminals are cesspools… Read Full Article
The holidays are a time for family, friends and celebration, but they can also be a time of stress, over-indulgence… Read Full Article
Getting up in the middle of the night to change wet sheets and console an embarrassed child happens in most… Read Full Article
If you are attempting to childproof your home, you likely know about outlet plugs. You have probably attached plastic locks… Read Full Article
It’s a battle that rarely has a victor. Nightly homework struggles can weigh heavy on both parents and children, leaving… Read Full Article
Parents take their newborns to the pediatrician six times during the first year of life. But the baby isn’t the… Read Full Article
It tends to strike when parents most need a break. At the end of the day, your well-fed, otherwise happy… Read Full Article
If a glowing blue light regularly emanates from beneath the door of one of your bedrooms late at night, there… Read Full Article
Germs! Whether you’re a germaphobe or someone who thinks the three-second rule is short by 20 minutes, we all know… Read Full Article
During the summer, we’re pretty good about following our young children around with water bottles, urging them to stay hydrated.… Read Full Article
Young children with maturing palates will often try their parents’ patience before they’ll try the food on their plates. If… Read Full Article
Your belly is growing … and so is your anxiety. Around the end of your second trimester, the realization suddenly… Read Full Article
As summer kicks off and your kids trade their books for boogie boards, it’s time to remember that no one… Read Full Article
Now that summer is in full swing, many of us will be spending more time at pools, lakes, beaches and… Read Full Article
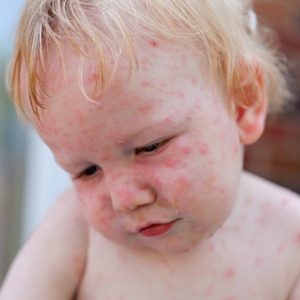
It’s not a very clever name for a disease, but it gets the point across. Hand, Foot, and Mouth (HFM)… Read Full Article
Earlier this month, the newly-expanded Hoag Health Center in Huntington Beach opened its doors to the public. Contributing another 50,000… Read Full Article
Bullying is any unwanted, repetitive, aggressive behavior due to a perceived power imbalance and can include verbal, physical, and/or social… Read Full Article
Tobacco smoke is a mixture of over 7,000 chemicals, hundreds of which are known toxins, 70 of which cause cancer.… Read Full Article
What is a well woman exam and why do you need one? The well woman exam is an annual physical… Read Full Article
One of the things I frequently hear from parents when discussing immunizations is a concern for the general safety of… Read Full Article
A D.O. is a doctor of osteopathy, also known as an “osteopath.” You may have seen these letters after a… Read Full Article
Very often I have patients who come in to my office concerned that they have an autoimmune condition such as… Read Full Article
If your child plays basketball, soccer, or some other winter sport, it may be time for yet another sports physical.… Read Full Article
“BRRRRRRIIINGGG!!!!” The alarm clock rings and I gaze drowsily towards the blurry clock. The bright red LED screen flashes “7:00AM”,… Read Full Article
Physical activity is important for all people and should be incorporated routinely in everyone’s life. Current recommendations regarding the type